Progams
NECTA Annual Drug Development Meeting
Each year we host our internationally attended flagship early phase drug development meeting. The goal of the meeting is to facilitate collaboration across academia, industry and government to accelerate early phase clinical trial work in Australia. To date, we can proudly point to excellent discussions and talks covering a range of emerging areas on oncological research. To find out more or partner with us, visit the dedicated website: www.drugdevelopment.com.au
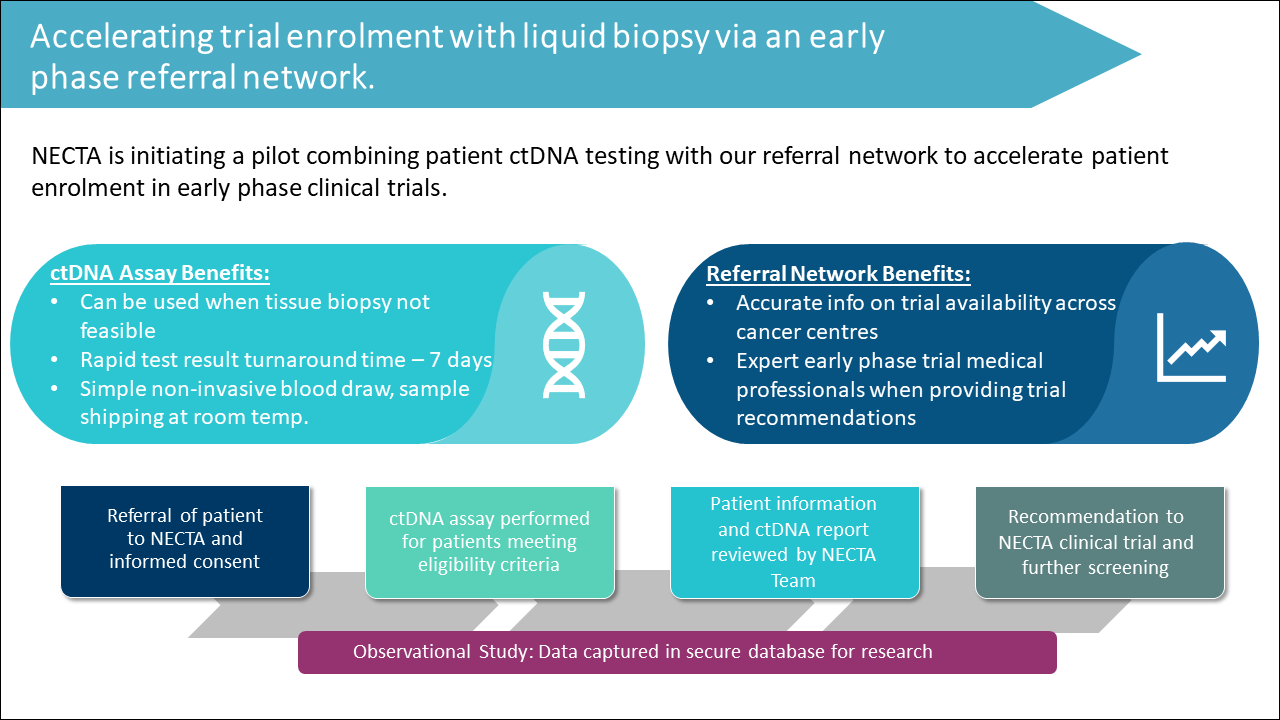
NECTA Referral Network with Liquid Biopsy
To assist in rapid and effective clinical trial placement, NECTA is pleased to announce the launch of a new initiative in 2022 evaluating the impact of our patient referral network in conjunction with liquid biopsy (circulating tumour DNA testing).
Currently, around one third of clinical trials at NECTA sites are biomarker related and so it is expected that using validated ctDNA testing will provide fast turnaround precision medicine recommendations for enrolment into early phase clinical trials. We have partnered to offer the next generation sequencing assay GUARDANT360® as the testing platform, which reports highly specific molecular results for review and recommendation by NECTA experts. Patient access outcomes will be reported and scientifically validated as an objective of an investigator initiated multicentre study.
This program is an early pilot and we are actively seeking further partnerships with sponsors to expand patient access to clinical trials with this platform.
For more information about how you can work with us, contact us here.
Currently, around one third of clinical trials at NECTA sites are biomarker related and so it is expected that using validated ctDNA testing will provide fast turnaround precision medicine recommendations for enrolment into early phase clinical trials. We have partnered to offer the next generation sequencing assay GUARDANT360® as the testing platform, which reports highly specific molecular results for review and recommendation by NECTA experts. Patient access outcomes will be reported and scientifically validated as an objective of an investigator initiated multicentre study.
This program is an early pilot and we are actively seeking further partnerships with sponsors to expand patient access to clinical trials with this platform.
For more information about how you can work with us, contact us here.
About GUARDANT360®
Personalised medicine for patients with advanced-stage cancer depends on identification of potentially actionable genetic alterations in the tumour. However, obtaining sufficient material from tumour biopsies is not always possible and liquid biopsies incorporating ctDNA have had utility in identifying actionable variants. Additionally, ctDNA profiling can determine the existing molecular alterations present in the tumour just prior to trial enrolment and overcomes the issue of tumour clonal evolution and heterogeneity compared with archival tissue samples.
Guardant 360 has a panel 74 clinically actionable cancer genes and utilises digital sequencing of ctDNA isolated from a simple, non-invasive blood draw. Distinct from other liquid biopsy assays, it has ultra-high specificity, detects single nucleotide variants, and copy number amplifications in panel genes including MSI-High. This test eliminates nearly all false positive results with individually tagged ctDNA molecules combined with post-sequencing bioinformatic reconstruction. The Guardant 360 test has the potential to evaluate the multiple genomic alterations cited in the United States National Comprehensive Cancer Network guidelines, and to prevent the time delays, cost and complications inherent in invasive biopsies.
Guardant 360 has a panel 74 clinically actionable cancer genes and utilises digital sequencing of ctDNA isolated from a simple, non-invasive blood draw. Distinct from other liquid biopsy assays, it has ultra-high specificity, detects single nucleotide variants, and copy number amplifications in panel genes including MSI-High. This test eliminates nearly all false positive results with individually tagged ctDNA molecules combined with post-sequencing bioinformatic reconstruction. The Guardant 360 test has the potential to evaluate the multiple genomic alterations cited in the United States National Comprehensive Cancer Network guidelines, and to prevent the time delays, cost and complications inherent in invasive biopsies.
NECTA and Guardant Health
Informational Webinar on Liquid Assays in Clinical Trial Enrolment
Presented by the New South Wales Early Phase Clinical Trials Alliance (NECTA) and Guardant Health AMEA, this webinar discusses the use of liquid biopsy in accelerating enrolment to early phase clinical trials for patients with advanced stage cancers.
Esteemed speakers from Australia, Israel, South Korea, and Singapore share their insights and experiences, while the webinar also explores:
The webinar can be viewed in the below video:
Esteemed speakers from Australia, Israel, South Korea, and Singapore share their insights and experiences, while the webinar also explores:
- Ways to improve screening and enrolment rates in early phase oncology clinical trials in Australia by leveraging a joint ctDNA testing initiative with NECTA and Guardant
- How to access large trial networks and services in Australia through NECTA
- Benefits of conducting clinical trials in Australia
The webinar can be viewed in the below video: